The process of starting pharmaceutical studies involves hundreds, sometimes thousands of forms and legal paperwork to be filled out. The role of an Independent Review Board (IRB) is to ensure that the accuracy, morality, and depth of these studies meet health and safety standards. Above all else, the role of an IRB is protect the people who volunteer as research subjects for studies.
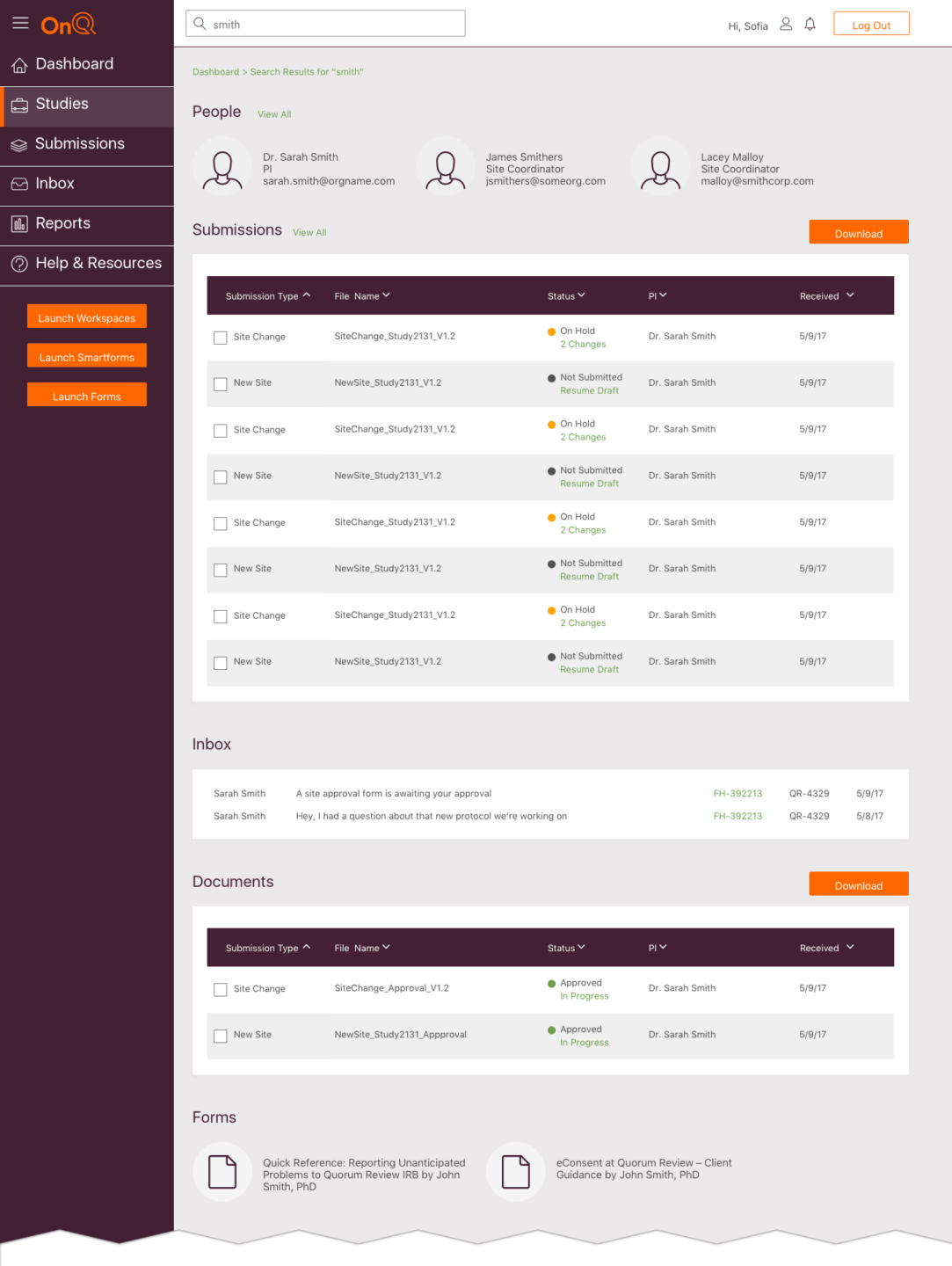
I designed a research study dashboard that connected board members, scientists, study managers and research teams for Quorum IRB. I participated in a three-day design thinking workshop with company stakeholders, customer advocates, engineers and researchers to plan the project, consolidate ideas, build momentum and get executive buy-in for the project.
After the workshop, I created personas, wrote user stories, conducted stakeholder interviews, researched competitors and participated in weekly collaborative review sessions with Quorum leadership and engineering teams to design and validate dashboard concepts.